Heavy Liquid for float sink separations
General information on heavy liquids
Comparison with other heavy liquids
General Information on Heavy Liquids
Heavy liquids are dense fluids or solutions used to separate materials of different density through their buoyancy. Materials with a density greater than the heavy liquid will sink, while materials with a density less than the heavy liquid will float on the liquid surface.
In the mineral industry, heavy liquids are commonly used in the laboratory to separate the “light” minerals such as quartz and clay from the “heavy” minerals. The density used for this type of separation is about 2.85 g/mL, nearly three times the density of water. Heavy liquid separations are carried out for varied reasons, depending on the industry. The mineral sands industry uses heavy liquid separations to check the grade of samples in their process, and to determine the efficiency of their industrial hydrocyclone separations. The diamond exploration industry uses heavy liquids to separate the dense indicator minerals from sand and clay.
Another use of heavy liquids is in paleontology. Typically, these heavy liquid separations are conducted at a lower density (e.g. 2.2 g/mL) since the separation is not between minerals of different types, but between fossil bones and minerals.
Organic heavy liquids
The organic heavy liquids form the "older" generation of heavy liquids. Two of the organic heavy liquids most commonly in use are bromoform and tetrabromoethane (TBE). Of these, bromoform has the lower viscosity (1.8 cP) but is considered more hazardous to work with because it has the higher vapour pressure (5.9 mm Hg at 25oC). TBE has a higher viscosity (9 cP) and a lower vapour pressure (0.02 mm Hg at 25oC).
Another organic heavy liquid, used when higher densities are required, is methylene iodide. Methylene iodide has a density of 3.31 g/mL, a vapour pressure of 1.2 mm Hg at 25oC, and a low viscosity of 2.6 cP.
The three commonly used organic heavy liquids all have problems with toxicity, and must be handled in a fume hood. Methylene iodide is moderately toxic by subcutaneous and other routes. Bromoform has been shown to cause severe liver damage and is poisonous by ingestion. There is evidence that it is a human mutagen and a neoplastigen on experimental animals, so it is potentially carcinogenic. TBE is poisonous by inhalation or ingestion and is a moderate irritant to the skin. It is a neoplastigen and mutagen with animals.
These liquids have the problem of being volatile and potentially causing health problems. For this reason, they are being phased out in the USA.
Tungsten based heavy liquids: SPT, LMT and LST
Two low-toxicity substitutes for the organic heavy liquids bromoform and tetrabromoethane (TBE) have been available for some time. They are SPT (sodium polytungstate, or sodium metatungstate), and the lithium equivalent LMT which is lithium metatungstate. These are all inorganic compounds, based on the [H2W12O40]6- polyanion, which is dissolved in water to form very dense solutions.
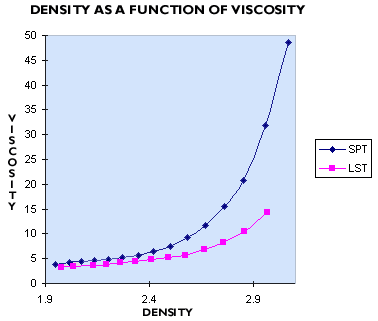
In our experience, SPT (sodium polytungstate) and LMT (lithium metatungstate) are not used by many mineral labs which need a heavy liquid with a density of 2.8 g/mL or more, because their solutions at these densities are too viscous. Typically the viscosity of these liquids is greater than 25 cP, or more than 25 times the viscosity of water. The higher viscosity of heavy liquids prepared from sodium metatungstate (also known as sodium polytungstate, or SPT) can be seen in the graph to the right (Figure 1). In practice, the higher viscosity of SPT leads to significantly slower and less efficient separations, as well as slow filtration.
Another disadvantage of SPT and LMT is their lack of thermal stability above 80oC. The lack of thermal stability of sodium metatungstate and lithium metatungstate means that solutions of SPT or LMT cannot be evaporated by boiling, so the recirculation and regeneration of these liquids requires special equipment, or is quite time consuming.
A more recent development is the low-toxicity heavy liquid called LST. LST Heavy Liquid has a low viscosity and excellent thermal stability. LST Heavy Liquid can be used up to a density of 2.9 g/mL at room temperature, and up to a density of 3.6 g/mL at elevated temperatures.
Physical Characteristics of LST Heavy Liquid
LST solids contain lithium heteropolytungstates in the form of extremely soluble, colourless hydrated crystals. Concentrated aqueous solutions of LST form colourless or pale yellow heavy liquids. LST Heavy Liquid is normally used with an aqueous base. However, the LST crystals are also highly soluble in acetone and alcohols, and can form dense liquids in these organic solvents. This provides a range of methods for the use and recycling of LST Heavy Liquid.
Thermal Stability of LST Heavy Liquid
LST Heavy Liquid maintained at a temperature of 140oC for over two weeks showed no observable change. The high thermal stability of LST heavy liquid allows it to be boiled to evaporate water and regain its original high density. The stability of LST Heavy Liquid means that recycling is rapid, simple and effective.
Operating Density of LST Heavy Liquid
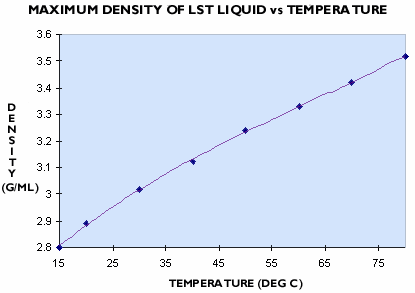
At 25oC LST Heavy Liquid can be used at any density up to 2.95 g/mL by diluting with water or by concentrating to remove water. The recommended operating density as a replacement for bromoform is 2.85 g/mL. At this density the viscosity is low, and the likelihood of crystallisation is reduced. Densities up to 3.6 g/mL can be achieved at higher temperatures. The maximum density of LST Heavy Liquid is given for a range of temperatures in Figure 2.
Viscosity of LST Heavy Liquid
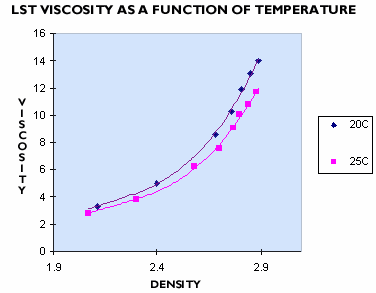
Low viscosity is a feature of LST Heavy Liquid. Grain separations in LST Heavy Liquid occur at about the same speed as when using TBE, but without the occupational hygiene risk of organohalides. The exact viscosity of the LST Heavy Liquid will vary with its temperature and density (see Figure 3), and is typically about 11 cP.
Storage and shelf life of LST Heavy Liquid
LST Heavy Liquid can be stored in closed plastic or glass containers. LST Heavy Liquid may cause corrosion of metals, so contact of LST with metals should be avoided during storage and heavy liquid separations.
Changes during Storage in Cold
If subject to very cold conditions, crystals may appear in LST Heavy Liquids. These will redissolve upon warming the heavy liquid for a few hours to 40oC, with occasional shaking or stirring.
Shelf Life
LST Heavy Liquid is a solution of stable inorganic ions in water, and has virtually an indefinite shelf life. In extended trials, LST Heavy Liquid which has been stored for over 10 years shows no sign of decomposition.
Recycling LST Heavy Liquid
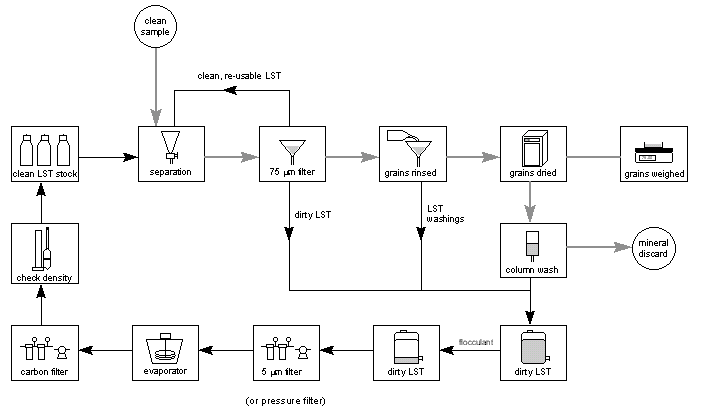
LST Heavy Liquid can be recovered from minerals quite easily by firstly washing the separated minerals with deionised water, filtering, and then concentrating the water washings by evaporation through boiling. The LST Heavy Liquid is not volatile so it is concentrated as the water evaporates. In this way, LST Heavy Liquid can be recycled with greater than 99% efficiency. The overall scheme for use in an industrial laboratory is shown in Figure 4.
The recovery of LST Heavy Liquid is high by this method. Typically, only 1.5 mL per kilogram of mineral sample of LST Heavy Liquid is lost with general usage.
SDS for LST Heavy Liquid
The Safety Data Sheet (SDS) for LST Heavy Liquid contains a more detailed summary of information from toxicity tests which have been conducted on LST Heavy Liquid. The SDS for LST Heavy Liquid can be downloaded as a pdf document.
Health & safety information: LST Heavy Liquid
LST Heavy Liquid was developed through an AMIRA research project. The driving force behind the development and introduction of LST Heavy Liquid was the need for a dense liquid to replace the undesirable dense organohalides such as tetrabromoethane (TBE) and bromoform. During the development of a suitable heavy liquid, safety was the prime consideration. Performance, i.e. high stability and low viscosity, was then optimised.
Detailed toxicity studies have been conducted on the components of LST Heavy Liquid in order to register the new substance for use in Australia. Worksafe approval for use in Australia was obtained in January 1996.
In 2000, Rio Tinto Exploration and Iluka Resources were awarded “Highly Commended” certificates in Australia’s National Safety and Health Innovation Awards for their part in supporting the use of LST Heavy Liquid to replace bromoform and TBE.
LST Heavy Liquids are of low toxicity. In this way they are similar to other liquids formed from the related polytungstate heavy liquid compounds, such as sodium polytungstate (sodium metatungstate) and lithium metatungstate.
Tests indicate that LST Heavy Liquid is unlikely to be a significant skin irritant. However, waterproof gloves should be worn while using it. The crystalline solids from LST Heavy Liquid are considered an eye hazard because of their potential irritancy when allowed to remain in contact with the eyes, and eye protection is required when using LST Heavy Liquid.
LST Heavy Liquids do not present any hazards from volatile vapours and do not require expensive fume extraction systems.